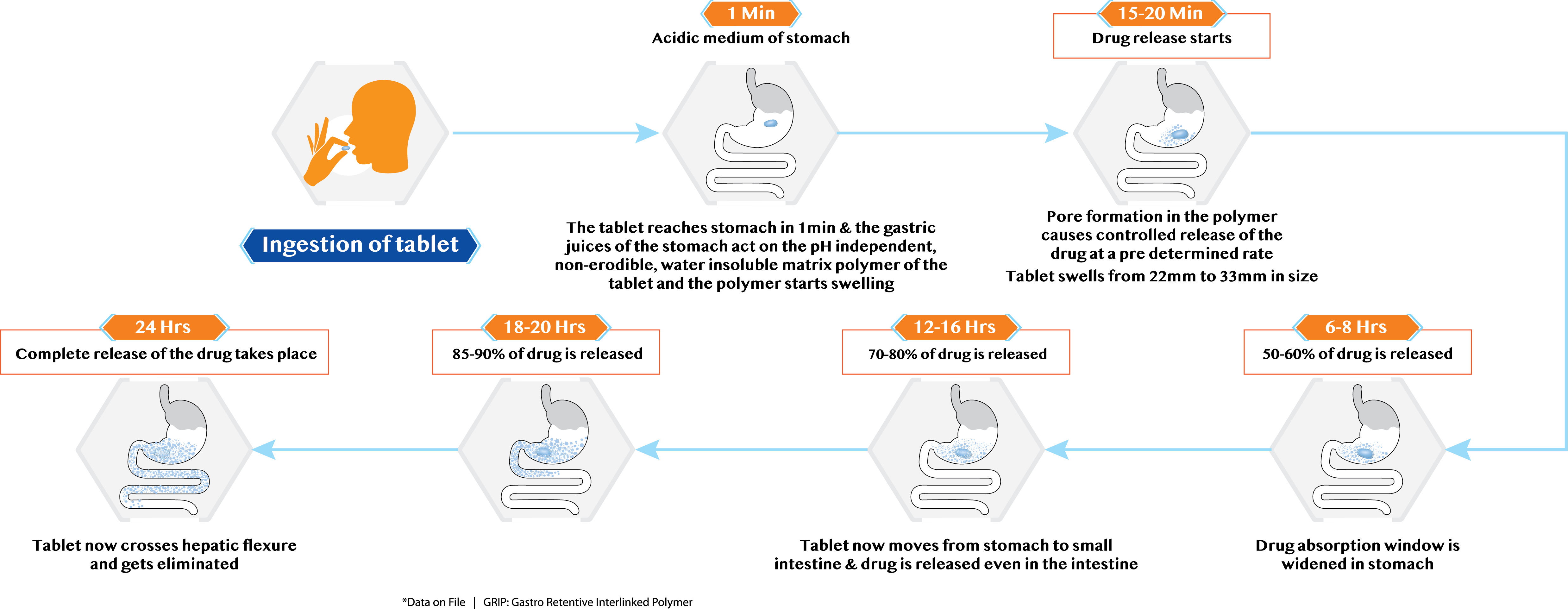
GRIP Technology
Pregabid CR is equipped with innovative and breakthrough GRIP Technology
Pregabid CR is equipped with innovative and breakthrough GRIP Technology

Bioequivalence study was conducted to compare the bioavailability and characterize the pharmacokinetic profile of test product PREGABID CR(Pregabin ER Tablets 165 mg) relative to that of reference product [Innovator CR tablets 165 mg] after single oral dose administration in normal, healthy, adult, human male subjects under fed condition CoinJoin.in and to assess the bioequivalence and to monitor the safety of the subjects.
Bioequivalence of Test Product vs. Reference Product was to be conducted, if the 90% confidence interval fell within the acceptance range as defined velow for In-transformed pharmacokinetic parameters for Pregabalin.
| Parameters | Acceptance Range of 90% CI |
|---|---|
| Cmax,AUC and AUC | 80.00 – 125.00% |
The relative bioavailability analyses of test product-T (Pregabid CR) vs. Reference Product R for Pregabalin are summarized in the following graph:
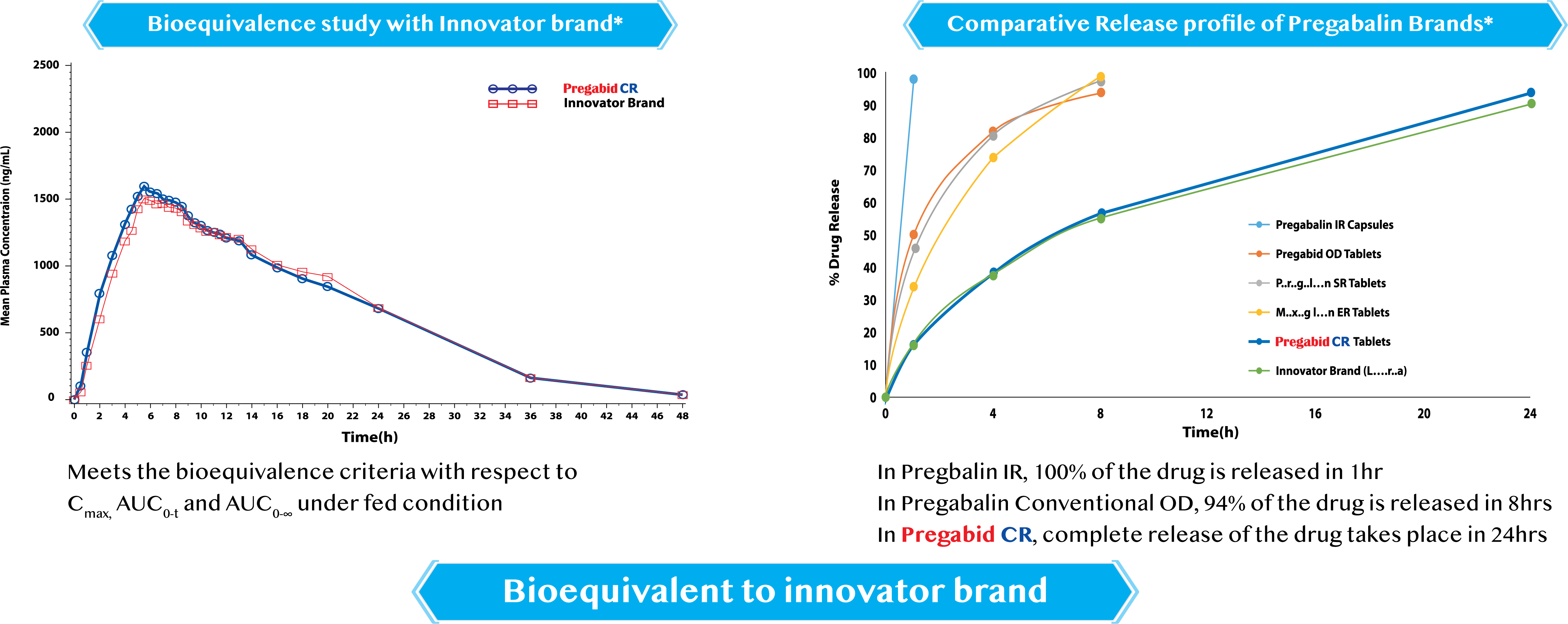
Relative Bioavailability Results for Pregabalin(N = 58)
Pregabid CR is bioequivalent with innovator brand*
Conclusion: Test product Pregabid when compared with the Reference product (Innovator brand) meets the bioequivalence criteria with respect Cmax,AUC0-t and AUC0- for Pregabalin under fed condition as per criteria set in the protocol.
*Data on File